Coordinate Covalent Bond Study Guide
INTRODUCTION
Football is a team sport. Imagine what will happen if a player doesn’t pass the ball to any other player on the team. The strength of a team resides in how well its players can cooperate and coordinate with one another. No matter how good an individual player is, the team’s stability depends on how well the ball is coordinated between different players. A similar case is with coordination bonds. To achieve stability in a molecule, some atoms share their lone pair of electrons with some other element.
WHAT IS A COORDINATE BOND?
A co-ordinate bond is an alternative covalent link produced when an electron pair from a single atom is shared. The very same atom provides both shared electrons. It’s also known as a dative bond. Coordinate covalent bonds are commonly produced in interactions involving two non-metals, such as hydrogen atoms, or when metal ions and ligands create bonds.
CHARACTERISTICS OF COORDINATE BOND
- The atom that exchanges an electron pair with itself is the donor in this sort of bonding.
- A receptor or acceptor is another atom that receives those shared pairs of electrons.
- An arrow pointing from the donor atom to the acceptor symbolizes the bond.
- Each atom achieves stability when the electron suffering is shared.
- The Lewis theory is based on this form of bonding.
- Studying co-ordinate covalent bonds will aid in the right construction of complex organic compounds.
COORDINATE BOND EXAMPLE
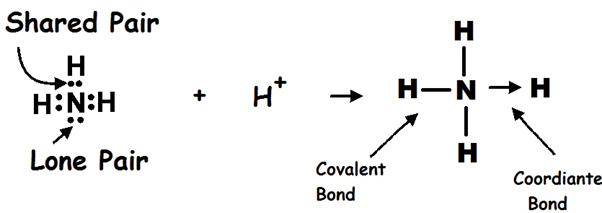
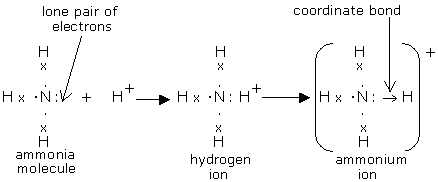
Ex.1 FORMATION OF AMMONIUM ION
The electron pair of the nitrogen atom in ammonia is donated to the vacant orbital of the H+ ion, making nitrogen the donor and H+ the acceptor, and a co-ordinate bond is created.
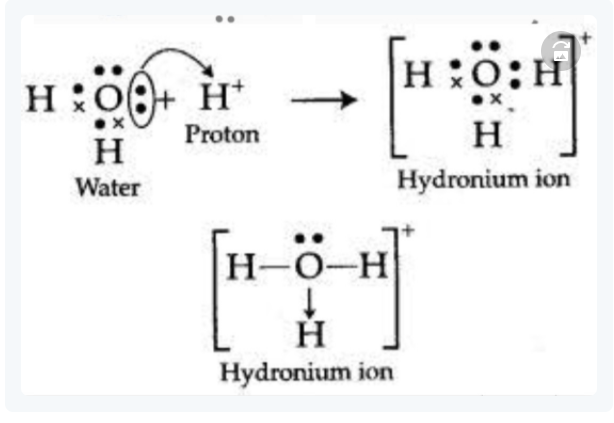
Ex.2 FORMATION OF HYDRONIUM ION
In water, an oxygen atom gives one electron pair to the empty orbital of the H+ ion, forming a dative connection. The donor atom is oxygen, while the acceptor atom is H+.
Formation of Ammonia Boron Trifluoride
The chemical formula for Boron Ammonia trifluoride, also known as trifluoroborane ammonia, is BF3H3N. It has an 84.84 u molecular weight. It has dative covalent bonding, which is another name for coordinate bonding. Both electrons contribute to a coordinate bond from the same atom. It is merely a one-sided sharing; the other atom does not share its electron. You may depict a dative bond by adding an arrowhead from the donor to the acceptor atom.
Properties of Coordinate Compounds
Covalent and ionic compounds have characteristics in common with coordinate compounds. A semipoloar coordinate bond exists.
- Their physical state: They are solids, liquids, and gases.
- Melting and boiling points: They have greater melting and boiling points than covalent compounds but lower values than ionic compounds.
- Solubility: They are readily soluble in non-polar solvents but sparingly soluble in polar solvents like water.
- Conductivity: Similar to covalent compounds, they are poor electricity conductors.
- Reactivity: They go through molecular reactions, which take a very long period to complete.
- Isomerism is the property of chemical species that have a molecular formula but are members of distinct compounds. They mainly display isomerism in complicated compounds.
- Stability: Because they share electrons naturally, they are quite stable.
- Dielectric Constant: They have high dielectric constant values.
CONCLUSION
- Coordinate covalent bonds can develop when one atom contributes a lone pair of electrons to the bonding.
- Coordinate covalent bonds are commonly produced in interactions involving two non-metals.
FAQs
1. How many bonds are in carbon monoxide?
Carbon Monoxide molecule contains a triple bond, two of which are covalent bonds, and one is a coordinate covalent bond.
2. What type of bond is CaO?
CaO has an ionic bond. The ionic bond is formed when one of the elements is metallic and the other is non-metallic. The cation is formed by Ca², and the anion is formed by O²-.
We hope you enjoyed studying this lesson and learned something cool about Coordinate Covalent Bond! Join our Discord community to get any questions you may have answered and to engage with other students just like you! Don’t forget to download our App to experience our fun, VR classrooms – we promise, it makes studying much more fun! 😎
]]>