Chemical Equilibrium Study Guide
INTRODUCTION
The word equilibrium literally means “balance.” Therefore, chemical equilibrium represents a state of balance in a chemical reaction. Chemical equilibrium is a state where there is no change in observable properties of the product involved in the process. Observable properties of a substance are its pressure, color, temperature, etc.
To put things in perspective, you can imagine melting ice at 0°C. In that state, both ice and water are present. However, melting of ice, dissolution of solute in solvent, etc., are examples of physical equilibrium. When a similar state is reached in a chemical reaction, it is termed a chemical equilibrium.
EQUILIBRIUM OF CHEMICAL CHANGE
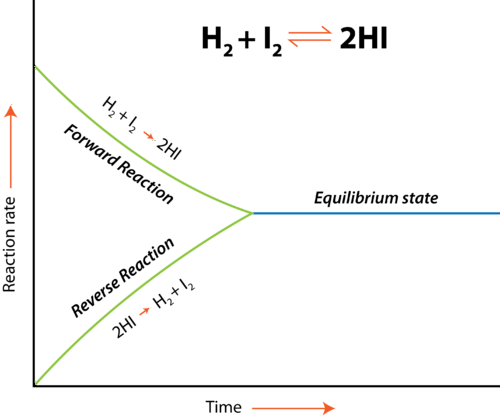
In a chemical reaction, equilibrium is achieved when the reaction is reversible. In cases as such, chemical equilibrium is defined as a state when the forward and backward reaction occurs at the same rate.
Alternatively, we may also define chemical equilibrium as a state where the volume of reactants and products does not change with time. In some cases, chemical equilibrium is also perceived as a stage where observable properties of the substances involved in the reaction do not change further with time when suitable conditions are provided.
Why is Chemical Equilibrium called Dynamic Equilibrium?
Chemical equilibrium is sometimes known as a dynamic equilibrium because it has the potential to move in favour of both the reactant and the product, a phenomenon known as equilibrium shifting. In a state of chemical equilibrium, however, the rate at which the reaction takes place is equal to the rate at which the product is produced. So, although it appears to be in equilibrium, a reaction is actually occurring. Therefore, dynamic equilibrium is in chemical equilibrium.
WHAT ARE THE CHARACTERISTICS OF CHEMICAL EQUILIBRIUM?
Let’s look at some of the properties of the equilibrium state:
- When equilibrium is achieved in a chemical reaction, the number of reactants and products becomes equal. Therefore, one can start the reaction from either end.
- When equilibrium is achieved at a given temperature, certain observable properties like pressure, density, color, etc., remain constant.
- In order to achieve equilibrium, the chemical reaction must be reversible and must be conducted in a closed space.
Types of Chemical Equilibrium
There are two types of chemical equilibrium:
• Homogeneous Equilibrium• Heterogeneous Equilibrium
Homogeneous Equilibrium
A chemical reaction is said to be in equilibrium when the rates of the forward and backward reactions are equal. Homogeneous and heterogeneous equilibrium are two other forms of equilibrium. A homogeneous mixture is a homogeneous equilibrium in one phase (products and reactants in a single solution).
Remember that in the equation, the reactants are on the left and the products are on the right. Thus, the solutes’ response is confined inside a single homogenous equilibrium. A chemical system with a heterogeneous equilibrium, on the other hand, contains reactants and products that are distributed throughout two or more phases.
Heterogeneous Equilibrium
When the reactants and products of an equilibrium reaction combine to produce a single phase, whether it be a gas or a liquid, the system is in homogeneous equilibrium. In such cases, there may be significant differences in the reactant and product concentrations. On the other hand, a heterogeneous equilibrium is a situation where the reactants, products, or both are present in more than one phase, for as when a gas reacts with a solid or liquid.
Factors Affecting Chemical Equilibrium
Temperature, pressure, and the system’s concentration are only a few of the variables that influence the equilibrium conditions.
Change in Concentration: The reaction that consumes the material introduced reduces the concentration of the reactants or products added.The reaction that is in the direction that replenishes the substance that is withdrawn relieves the concentration of reactants or products eliminated.In a chemical equilibrium, the composition of the mixture changes as the reactant or product concentration is altered.
Change in Pressure: The volume change is what causes the change in pressure. Since the total quantity of gaseous reactants and products has changed as a result of the change in pressure, the gaseous reaction may be impacted. Le Chatelier’s principle states that the volume is independent of pressure in heterogeneous chemical equilibrium, hence the change in pressure in both liquids and solids may be disregarded.
Temperature Change: Le Chatelier’s Principle governs how temperature affects chemical equilibrium and is dependent on the reaction’s ΔH sign. An exothermic reaction’s equilibrium constant lowers with rising temperature.The equilibrium constant of an endothermic process rises as the temperature rises.
__Catalyst Effect: __ A catalyst has no impact on the chemical equilibrium. It just quickens a response. In actuality, a catalyst quickens both the forward and the backward response. As a result, the reaction reaches equilibrium more quickly. In a catalyzed or non-catalyzed reaction, the same number of reactants and products will be present at equilibrium.
Examples of Chemical Equilibrium
In chemical processes, the forward reaction transforms reactants into products, while the reverse reaction transforms products into reactants.
The pace of the forward and backward responses may become equal after some time has passed after the reaction began. Following this, the number of reactants converted will be generated once again by the reverse reaction, preventing any further changes in the concentration of reactants and products.
PCl5 ⇌ PCl3 +PCl2
Importance of Chemical Equilibrium
It is helpful in several industrial operations, including• Ammonia is made using Haber’s method, which produces more ammonia at low temperatures, high pressures, and in the presence of iron as a catalyst. Nitrogen and hydrogen combine to make ammonia during this process.• Sulfuric acid is made through a contact process: The primary reaction in this process is the oxidation of sulfur dioxide into sulfur trioxide. Equilibrium in chemistry is involved here.
CONCLUSION
- Equilibrium means a state of balance.
- In physical equilibrium, two states of matter can exist simultaneously. For example, melting of ice at 0 degrees Celsius.
- Chemical equilibrium can be loosely defined as a state in a chemical reaction when there is no change in the observable properties.
- Chemical equilibrium can be defined as a state in a reversible reaction where the rate of forward and backward reaction becomes equal.
- We may also define chemical equilibrium as a state where there is no further change in the volume of reactants and products with time.
- Lastly, chemical equilibrium is also said to be achieved when there is no observable change in the properties of the substances involved in the process.
- To identify the equilibrium stage, take note of three characteristics:
- An equal volume of reactants and products.
- No change in observable properties with time.
- The reversible reaction is carried out in closed space.
FAQs
1. What is an equilibrium crash course?
A crash course on chemical equilibrium is a lesson plan that covers all the aspects of equilibrium briefly. The course mostly has an equilibrium video so that you can be exam ready within minutes.
2. How do you explain equilibrium in chemistry?
Chemical equilibrium is a state in a reversible chemical reaction when there is no change in the observable properties with time. For further details, you can join a chemistry crash course that will prepare you for your upcoming exams.
We hope you enjoyed studying this lesson and learned something cool about Chemical Equilibrium! Join our Discord community to get any questions you may have answered and to engage with other students just like you! Don’t forget to download our App to experience our fun VR classrooms – we promise it makes studying much more fun! 😎
]]>