Properties of Biological Macromolecules Study Guide
Introduction:
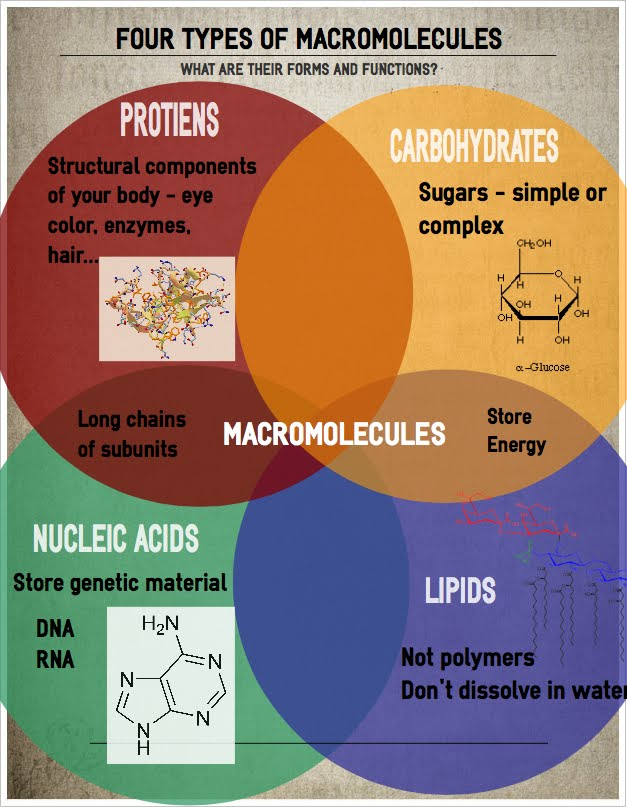
Biomolecules, also called biological molecules, are substances produced by cells and living organisms. Biomolecules have a wide range of sizes and structures and perform a vast array of functions. The four major types of macromolecules are carbohydrates, lipids, nucleic acids, and proteins.
Structure and functions of polymers:
The structure and functions of Polymers are influenced by the way monomers react or join together to form a polymer. The presence of strong intermolecular forces impacts the unique physical properties of polymers like density, crystallinity, melting point, and strength.
- Carbohydrates: Carbohydrates are made up of sugar monomers whose structures determine the properties and functions of the molecules.
- Proteins: The specific order of amino acids in a polypeptide determines the overall shape of the protein
- Nucleic acids: Biological information is encoded in sequences of nucleotide monomers in nucleic acids. Each nucleotide is made up of its structural components: ∘ a five-carbon sugar (Deoxyribose or Ribose) ∘ a Phosphate ∘ a Nitrogen base (adenine, thymine, guanine, cytosine, or uracil)
- Lipids: Differences in saturation determine the structure and function of the lipids.
Biological macromolecules
Biological macromolecules are very large molecules formed due to the polymerization of smaller molecules called monomers.
Carbohydrates
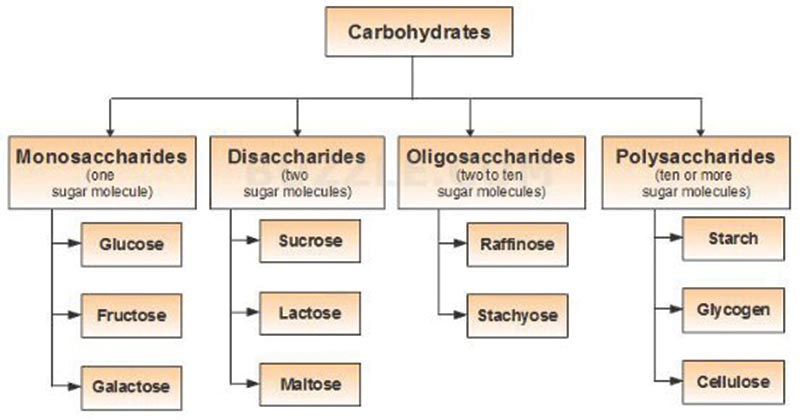
Carbohydrates are biological molecules formed from carbon, hydrogen, and oxygen, in a ratio of approximately one carbon atom to one water molecule. Carbohydrates get their name from this composition, and it is made up of carbon plus water. Biologically essential carbohydrates can be categorized into three categories:
a) Monosaccharide: The most common example is glucose.
Physical properties of monosaccharides are:
- Monosaccharides are colorless and crystalline compounds.
- They are readily soluble in water.
- They have a sweet taste.
Chemical properties of monosaccharides are:
Monosaccharides are polyhydroxy aldehydes or Ketones – they are molecules with more than one hydroxyl group (―OH) and a carbonyl group (C=O) either at the terminal carbon atom (aldose) or at the second carbon atom (ketose).
b) Disaccharides: It is produced when two monosaccharides undergo a dehydration reaction.
Physical properties of disaccharides are:
- Crystalline
- Water-soluble
- Sweet to taste
- They are broken into monosaccharides before being absorbed and used for energy
c) Polysaccharides: A long chain of monosaccharides connected by Glycosidic bonds are called polysaccharides.
The following chemical properties characterize polysaccharides:
- Not sweet
- Many are insoluble in water
- Do not form crystals when desiccated
- Compact and not osmotically active inside the cells
- Can be extracted to create a white powder
Lipids
Oils and fats, which may be saturated or unsaturated, are healthy and serve essential plants and animal functions. Lipids don’t make polymers.
Similar to carbohydrates, lipids are made up of carbon, hydrogen, and oxygen, but the atoms are arranged differently. Most lipids are hydrophobic and nonpolar. Major lipid types are Fats and Oils; Waxes, Phospholipids, and Steroids.
Fat normally comprises three fatty acids bonded to one molecule of glycerol, forming triglycerides. The fatty acids may be saturated or unsaturated, depending on the presence or absence of double bonds in the hydrocarbon chain. A saturated fatty acid has the maximum number of hydrogen atoms bonded to carbon. Generally, fats in a liquid state at room temperature tend to be more unsaturated than fats that are solid state at room temperature (Eg: Canola oil).
The properties of lipids are:
- They have minimal or no affinity for water because it consists of mostly hydrocarbons.
- Highly diverse in function and form. Fats store a large amount of energy.
Phospholipids are a special type of lipid-linked with cell membranes and normally have a glycerol backbone, to which two fatty acid chains and a phosphate-containing group are attached. Due to this, phospholipids are considered amphipathic because they have both hydrophobic and hydrophilic components.
Steroids are classified as lipids based on their hydrophobic properties, although the molecular structures of steroids differ from that of triglycerides and phospholipids. Cholesterol is a form of steroid in animal cells’ plasma membrane. Cholesterol is also the forerunner of steroid hormones such as testosterone.
Proteins:
The protein enzymes function as catalysts in cells and perform adaptable metabolism by selectively accelerating chemical reactions without consuming them. Proteins are polymers of amino acids.
Physical properties of proteins include:
- They are colorless and usually tasteless
- Their shape varies from simple crystalloid spherical structures to long fibrillar structures
- have large molecular weights
- Amphoteric Nature: Proteins are amphoteric like amino acids (i.e., they act both as acids and alkalis)
- Ion Binding Capacity: Proteins form salts with both anions and cations, based on their net charge
- Solubility: The solubility of proteins is influenced by pH
Chemical Properties of proteins are:
- Hydrolysis: Proteins are hydrolyzed by several hydrolytic agents
- When free amino acids or proteins are treated with Hydrochloric acid, the acid salts are formed
Nucleic Acids:
The biochemical properties of nucleic acids are:
- Ultraviolet absorption: The extent to which DNA is single-stranded or double-stranded can be determined by monitoring UV absorption
- Chemical modification: DNA can also be cleaved and degraded by enzymes called nucleases
- Mutation: Chemical modification of DNA can lead to mutations in the genetic material.
Conclusion:
- Biological macromolecules have a wide range of sizes and structures, and they perform several functions.
- The four major types of macromolecules are carbohydrates, lipids, nucleic acids, and proteins.
- The properties of these macromolecules influence their functions, and therefore applications in life.
FAQs:
1. What are the four types of macromolecules and their properties and functions?
The four major macromolecules and their respective functions are:
- Nucleic acids: Stores and transfers genetic info.
- Carbohydrates: Store energy, provide fuel, and build structure in the body; the main source of energy, the structure of the plant cell wall.
- Lipid: Acts as an Insulator and stores fat and energy.
- Protein: Provide structural support, transport, enzymes, movement, and defense.
2. How do the structures of biological macromolecules determine their properties and functions?
The three-dimensional structure of biological macromolecules determines how they function. The three-dimensional shape of proteins and nucleic acids endows them with their biological activities.
3. What is the structure of biological macromolecules?
Biological macromolecules are large and complex and are formed by the polymerization of smaller units called monomers. Macromolecules include Proteins, Nucleic acids, Carbohydrates, and Lipids.
4. What is a biological macromolecule?
Biological macromolecules are very large molecules formed due to the polymerization of smaller molecules called monomers.
5. What is the importance of biological macromolecules to our lives?
The importance of biological macromolecules to our lives include:
- Providing structural support
- Being a source of stored fuel
- Storing and retrieving genetic information
- Speeding or hastening biochemical reactions
We hope you enjoyed studying this lesson and learned something cool about the Properties of Biological Macromolecules! Join our Discord community to get any questions you may have answered and to engage with other students just like you! Don’t forget to download our App and check out our fun VR classrooms – we promise, it makes studying much more fun 😎
Sources:
- Biomolecules https://ncert.nic.in/textbook/pdf/kebo109.pdf Accessed on 29 Dec, 2021
- Properties of Biological Macromolecules https://www.britannica.com/science/nucleic-acid/Methylation Accessed on 29 Dec, 2021
- Biological Macromolecules https://alevelbiology.co.uk/notes/proteins-physical-chemical-properties/ Accessed on 29 Dec, 2021
- Biological Macromolecules https://biologydictionary.net/ap-biology/1-4-properties-of-biological-macromolecules/ Accessed on 29 Dec, 2021